 The attraction between positively charged nucleus and negatively charged electrons keeps the electrons within the atom.Negatively charged electrons rotate around the nucleus in orbits (or shells) The convention of indicating the atomic number and mass number of an element Atomic number.A = Z + N is called the atomic mass number.The nucleus contains N number of chargeless neutrons.The nucleus contains Z number of positively charged protons.Symbol of the element A X Mass number Z 40 Ca 23 Na 1 H 20 11 1 Protons = ? Protons = ? Protons = ? Electrons = ? Electrons = ? Electrons = ? Neutrons = ? Neutrons = ? Neutrons = ? Inside the atomThe fundamental particles Part 1: Atomic structure (contd) Inside the atom Negatively charged electrons orbiting around extremely small positively charged solid nucleus Bohr (1913)Electrons can occupy only ‘allowed’ orbits having stable energy levels Progressive changes in the understanding of the atom The scientist Niels Bohr further improved this model by stating that electrons can only occupy ‘allowed’ orbits whose energy levels are stableĭalton (1808) The smallest indivisible particle of an element Thomson (1897) negatively charged electrons embedded in a positively charged solid sphere Rutherford (1911) Planetary model. 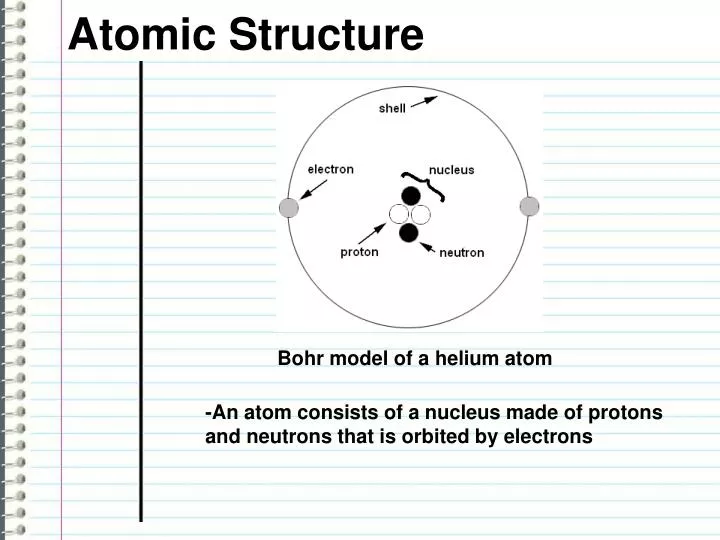
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
